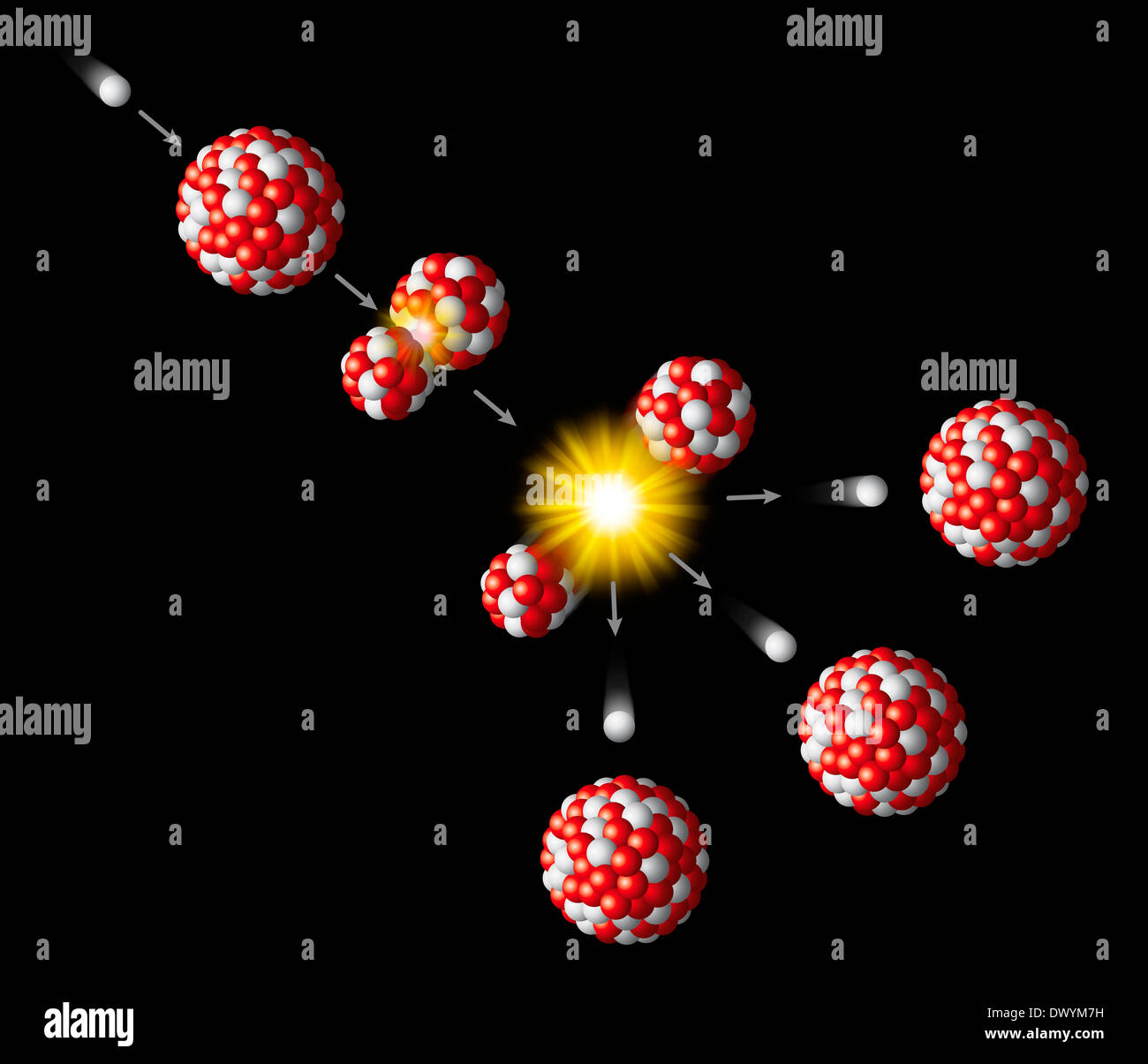
It is of further concern in the human body because iodine is quickly swept up by the thyroid, so that the total intake of iodine becomes concentrated there. Iodine-131 is a major concern in any kind of radiation release from a nuclear accident because it is volatile and because it is highly radioactive, having an 8 day half-life. Wiki: Strontium-90 Strontium nuclear dataĪn inevitable byproduct of nuclear fission is the production of fission products which are highly radioactive. Strontium-90 undergoes beta decay, emitting electrons with energy 0.546 MeV with a half-life of 28.8 years. Strontium-90 is not quite as likely as cesium-137 to be released as a part of a nuclear reactor accident because it is much less volatile, but is probably the most dangerous component of the radioactive fallout from a nuclear weapon. It has the potential for causing cancer or damaging the rapidly reproducing bone marrow cells. As a part of the bones, it is not subsequently excreted like cesium-137 would be. Strontium-90 mimics the properties of calcium and is taken up by living organisms and made a part of their electrolytes as well as deposited in bones. It ensures that they are not only highly radioactive but also have a long enough halflife to be around for hundreds of years. They both have intermediate halflives of around 30 years, which is the worst range for half-lives of radioactive contaminants. Strontium-90 and cesium-137 are the radioisotopes which should be most closely guarded against release into the environment. 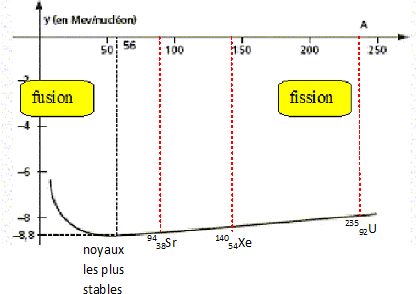
This ensures that cesium as a contaminant will be ingested, because potassium is needed by all living things.Įnvironmental Protection Agency bulletin.Īn inevitable byproduct of nuclear fission is the production of fission products which are highly radioactive. The gamma radiation is very penetrating, and the beta radiation, though very short range, is very dangerous when ingested because it deposits all that energy in a very short distance in tissue.Ĭesium's danger as an environmental hazard, damaging when ingested, is made worse by its mimicing of potassium's chemical properties. Both the electron and gamma emissions are highly ionizing radiation. Cesium-137 decay has a half-life of 30.07 years and proceeds by both beta decay and gamma emission from an intermediate state. This means that it is passed on up the food chain and reconcentrated from the environment by that process. Besides its persistence and high activity, cesium-137 has the further insidious property of being mistaken for potassium by living organisms and taken up as part of the fluid electrolytes. Iodine-131 may give a higher initial dose, but its short halflife of 8 days ensures that it will soon be gone. Their intermediate half-lives of about 30 years suggests that they are not only highly radioactive but that they have a long enough halflife to be around for hundreds of years. Cesium-137 and strontium-90 are the most dangerous radioisotopes to the environment in terms of their long-term effects. This particular set of fragments from uranium-235 fission undergoes a series of beta decays to form stable end products.Īn inevitable byproduct of nuclear fission is the production of fission products which are highly radioactive. These fragments are not so dangerous as intermediate half-life fragments such as cesium-137. Strontium-94 decays with a half-life of 75 seconds, finally producing the stable isotope zirconium-94. Highly radioactive, the xenon decays with a half-life of 14 seconds and finally produces the stable isotope cerium-140. Show exampleĪ common pair of fragments from uranium-235 fission is xenon and strontium: 
Most of these fission fragments are highly unstable ( radioactive), and some of them such as cesium-137 and strontium-90 are extremely dangerous when released to the environment. It is much more probable to break up into unequal fragments, and the most probable fragment masses are around mass 95 and 137. When uranium-235 undergoes fission, the average of the fragment mass is about 118, but very few fragments near that average are found. Nuclear Fission Fragments Fission Fragments
0 Comments
Leave a Reply. |
 RSS Feed
RSS Feed
